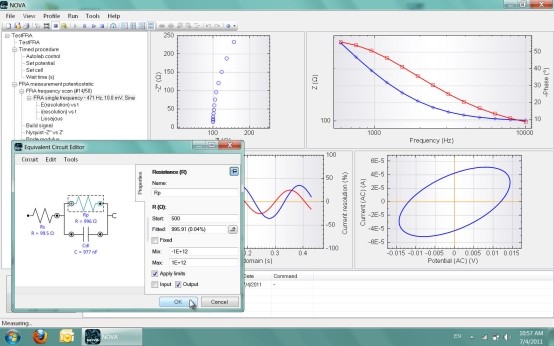
Previous studies indicated that monomers containing aromatic groups that are directly bonded to oxygen are easier to polymerize. An example of a non-conducting polymer is the thin electropolymerised film of poly, which can be used to eliminate electrochemical interference from ascorbate, urea, acetaminophen and other oxidizable species. Furthermore, non-conducting films are well suited for capacitance measurements where the electrode surface needs to be properly insulated. Second, the non-conducting polymer films are generally found to be more effective in both preventing the biosensor from fouling and eliminating the interference from electroactive species, such as ascorbic acid and uric acid. Biosensors prepared in this way generally have the advantages of fast response and high sensitivity because of relatively high enzyme loading. The biosensors based on immobilizing enzymes in non-conducting films have some advantages over conducting films: First, the film thickness of the non-conducting polymer is self-controlled during electropolymerisation, and a very thin and uniform film can be obtained. Also, the thickness of the electropolymerised film and the amount of immobilized enzyme can be controlled easily during electropolymerisation.Īfter conducting polymers, non-conducting polymers are emerging as a novel support matrix for the immobilization of biomolecules because they offer impressive advantages, including excellent perm selectivity and high reproducibility, in addition to most of the reported merits of conducting polymers. This is because these polymers have a high conductivity and stability in both air and aqueous solution. Polythionine and polythiophene, has been studied extensively for the development of biosensors. Electropolymerisation of conducting polymers, such as polypyrrole (PPy), polyaniline, polyacetylene, polyindole, The use of nanoscale polymer films permits better selectivity and faster measurements and has stimulated the development of new polymer films of varied chemical natures. Electrodes coated with polymeric films containing reactive groups can be connected to a cell so that they function as working electrodes, leading to the desired organic substrate transformations. Modified electrodes are useful because they can be applied to several electrochemical systems without modification of their characteristic or efficiency, thus enabling their reuse. The polymer film coated on the transducer surface for the immobilization of biomaterial must be able to efficiently spread the electrical potential produced by the biochemical reaction to the transducer, to ensure reproducibility and to amplify the signal. Polymer-modified electrodes, built up by deposition of electroactive polymeric films on conductive substrates, have been a major area of research for more than two decades. Keywords Surface modification polytyramine impedance biosensor tyramine cyclic voltammetry. Electrodes modified with polytyramine can be also fundamental to construct biosensors by enzymes immobilization at its surface. The voltammetric and impedance studies showed that polytyramine layer could be employed to determine biologically active substances. The resulting polytyramine film and particles were characterized through scanning electron microscopy, FTIR, cyclic voltammetry and impedance spectroscopy. The modified electrodes exhibited a good sensitivity, reproducibility, and stability. The resulting particles were deposited onto gold electrode through drop casting method.

Similarly a new approach was used to chemically polymerize tyramine in an alkaline media using FeCl3 as an oxidant. The electrodeposition was carried out in 0.025M tyramine solution in 0.3 M NaOH in methanol at the voltage ranging from 0V to 1.6 V at the scan rate of 100 mVs-1. The aim of the studies was to modify surface of gold electrode with the layer of polytyramine synthesized through chemical and electrochemical methods. Modifying layers allow scientists to build electrodes with required properties such as for example: selectivity, stability, precision, durability, range of potentials etc. Professor of Colour & Polymer Science University of LeedsĪbstract Developing modified electrodes through electrodepositing conductive polymer layer has been continuous studied in areas such as technology, electronics, medicine, biology and many others. Wajiha Abbasi Department of Chemistry, University of Leeds Electrode Modification Through Chemical and Electrochemical Deposition of Polytyramine Film for Biosensing Applicationĭr.

